Page 18 - Application Notebook - Solution for Food Development
P. 18
LAAN-A-LC-E240
Application High Performance Liquid Chromatography
News Analysis of Histamine and Tyramine Using Prominence
Amino Acid Analysis System
No.L463
Putrefactive non-volatile amines, histamine and tyramine, are mV
formed through decomposition of histidine and tyrosine, 200
respectively, due to the action of microorganisms. When 1 ■Peaks
ingested food such as processed products and red-fleshed fish 1. Histamine
such as tuna, bonito, mackerel, etc., contain a large amount of 150 2. Tyramine
histamine, food poisoning symptoms such as fever, hives, and
palpitations may appear. There are also reported cases of food 100
poisoning associated with fermented foods such as wine and
cheese. Further, tyramine can also strengthen the toxicity of 2
histamine, and has been reported as a causative agent in food- 50
associated migraine.
Although there are no specific histamine-related regulations in
Japan, in other countries, including the United States and the 0
EU, Codex (International Food Standards) regulatory limits for 0 5 10 15 min
histamine have been established for fish and fishery products.
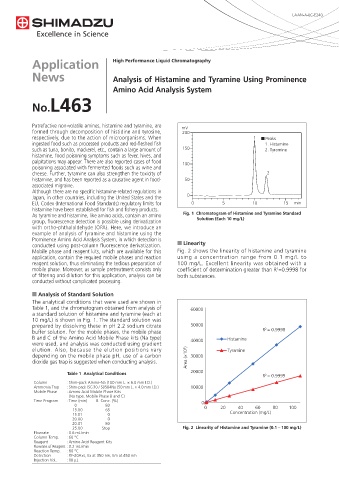
As tyramine and histamine, like amino acids, contain an amino Fig. 1 Chromatogram of Histamine and Tyramine Standard
group, fluorescence detection is possible using derivatization Solution (Each 10 mg/L)
with ortho-phthalaldehyde (OPA). Here, we introduce an
example of analysis of tyramine and histamine using the
Prominence Amino Acid Analysis System, in which detection is
conducted using post-column fluorescence derivatization. n Linearity
Mobile phase and reagent kits, which are available for this Fig. 2 shows the linearity of histamine and tyramine
application, contain the required mobile phases and reaction using a concentration range from 0.1 mg/L to
reagent solution, thus eliminating the tedious preparation of 100 mg/L. Excellent linearity was obtained with a
mobile phase. Moreover, as sample pretreatment consists only coefficient of determination greater than R =0.9998 for
2
of filtering and dilution for this application, analysis can be both substances.
conducted without complicated processing.
n Analysis of Standard Solution
The analytical conditions that were used are shown in
Table 1, and the chromatogram obtained from analysis of 60000
a standard solution of histamine and tyramine (each at
10 mg/L) is shown in Fig. 1. The standard solution was
prepared by dissolving these in pH 2.2 sodium citrate 50000
buffer solution. For the mobile phases, the mobile phase R 2 = 0.9998
B and C of the Amino Acid Mobile Phase kits (Na type) 40000 Histamine
were used, and analysis was conducted using gradient
elution. Also, because the elution positions vary Tyramine
depending on the mobile phase pH, use of a carbon Area (×10 3 ) 30000
dioxide gas trap is suggested when conducting analysis.
20000
Table 1 Analytical Conditions R 2 = 0.9999
Column : Shim-pack Amino-Na (100 mm L. × 6.0 mm I.D.)
Ammonia Trap : Shim-pack ISC-30 / S0504Na (50 mm L. × 4.0 mm I.D.) 10000
Mobile Phase : Amino Acid Mobile Phase Kits
(Na type, Mobile Phase B and C)
Time Program : Time (min) B. Conc. (%)
0 80 0 0 20 40 60 80 100
15.00 65
15.01 0 Concentration (mg/L)
20.00 0
20.01 80
25.00 Stop Fig. 2 Linearity of Histamine and Tyramine (0.1 – 100 mg/L)
Flowrate : 0.6 mL/min
Column Temp. : 60 °C
Reagent : Amino Acid Reagent Kits
Flowrate of Reagent : 0.2 mL/min
Reaction Temp. : 60 °C
Detection : RF-20Axs, Ex at 350 nm, Em at 450 nm
Injection Vol. : 10 µL