Page 106 - Application Handbook - Liquid Chromatography
P. 106
LAAN-A-LC-E280
Application High Performance Liquid Chromatography
News Analysis of Ions in Drugs (Part 5)
Analysis of Organic Acid Counterions by Ion Exclusion
No.L504 Chromatography
The physicochemical and pharmacokinetic properties of The number of columns (i.e., the size of the separating
active pharmaceutical ingredients change depending on space) and column temperature are important factors in
the counterion used, and during drug development the separation of multi-component mixtures of organic
various counterions are tested to select the optimum acids using ion exclusion chromatography.
salt. Consequently, two columns and a column temperature
Residual inorganic impurities from catalysts or ions used of 50 ˚C were used for the analysis of formic acid,
during synthesis can also affect product solubility and acetic acid, fumaric acid, and maleic acid.
stability, so it is extremely important that ion impurities
are identified.
Application News No. L457 described an example
analysis of chloride, formic acid, acetic acid, and Table 1 Analytical Conditions
trifluoroacetic acid ions present in drugs using ion Column : Shim-pack SCR-102H
2 columns (300 mm L. × 8.0 mm I.D.)
chromatography. As mentioned above, drugs can Mobile Phase : 5.0 mmol/L Perchloric Acid aq. solution
contain multiple different counterions and it is Flowrate : 0.8 mL/min
sometimes difficult to separate completely the peaks of Column Temp. : 50 ˚C
these ions using ion chromatography. Injection Volume : 10 μL
We describe an example analysis that uses ion exclusion Detection : UV-VIS detector (SPD-20A) at 210 nm
chromatography to increase separation selectivity for
formic acid, acetic acid, fumaric acid, and maleic acid,
which are organic acids frequently used for drug
counterions.
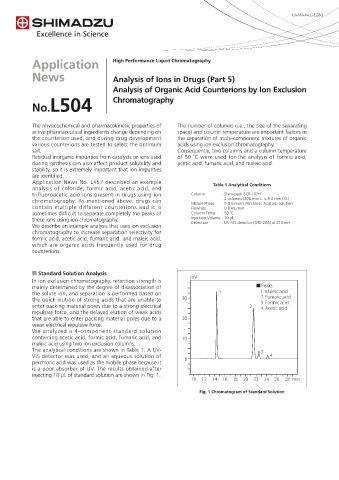
n Standard Solution Analysis
In ion exclusion chromatography, retention strength is mV
mainly determined by the degree of disassociation of ■Peaks
the solute ion, and separation is performed based on 1 1. Maleic acid
the quick elution of strong acids that are unable to 30 2. Fumaric acid
3. Formic acid
enter packing material pores due to a strong electrical 2 4. Acetic acid
repulsive force, and the delayed elution of weak acids
that are able to enter packing material pores due to a 20
weak electrical repulsive force.
We analyzed a 4-component standard solution
containing acetic acid, formic acid, fumaric acid, and 10
maleic acid using two ion exclusion columns.
The analytical conditions are shown in Table 1. A UV- 3
VIS detector was used, and an aqueous solution of 4
perchloric acid was used as the mobile phase because it 0
is a poor absorber of UV. The results obtained after
injecting 10 μL of standard solution are shown in Fig. 1.
10 12 14 16 18 20 22 24 26 28 min
Fig. 1 Chromatogram of Standard Solution